.png)
• Designed as a packaging solution for sterilization applications, the Medguard SafeCheck Tyvek® Sterilization Reel is a microbial barrier system that ensures the sterility of the packed medical device is safely maintained from the moment of sterilization until the time of use.
• The sterilization reel is used for low-temperature Hydrogen Peroxide (H₂O₂) sterilization methods.
• It is preferred for low-temperature sterilization due to its porous structure.
• The reel meets the high standards of quality, hygiene, and safety required in the medical industry.
• The sterilization reel is made of PET/PE transparent copolymer film and uncoated Tyvek® web.
• It features superior microbial barrier properties.
• It offers a suitable packaging solution for your medical device with its wide range of sizes.
• The process indicator does not contain lead, heavy metals, or other chemicals harmful to health.
• The product meets the performance requirements specified in ISO 11607, ISO 11140-1, and EN 868-9 standards.
• The LOT number, Production Date, and Expiry Date are indicated on the product label.
• The shelf life of the product is 5 years.
• Tyvek® is a registered trademark of DuPont™.
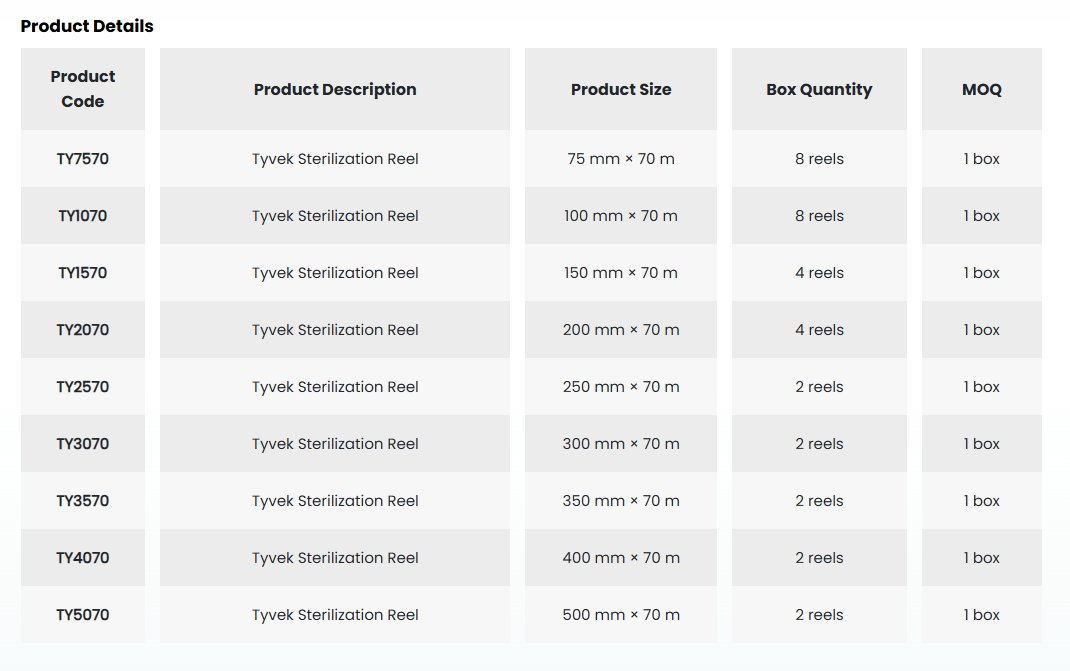

• Designed as a packaging solution for sterilization applications, the Medguard SafeCheck Sterilization Reel is a microbial barrier system that ensures the sterility of the packed medical device is safely maintained from the moment of sterilization until the time of use.
• It offers a suitable packaging solution for your medical device with a wide range of available sizes.
• It features superior barrier properties.
• The product is made of PET/PP copolymer film and 60 GSM or 70 GSM medical kraft paper.
• Steam, Ethylene Oxide (EO), and Formaldehyde (FO) indicator printing options are available.
• The process indicator does not contain lead, heavy metals, or other chemicals harmful thealth.
• The product meets the performance requirements specified in IS11607, IS11140-1, and EN 868-5 standards.
• LOT number, Production Date, and Expiry Date are indicated on the product label.
• The shelf life of the product is 5 years.
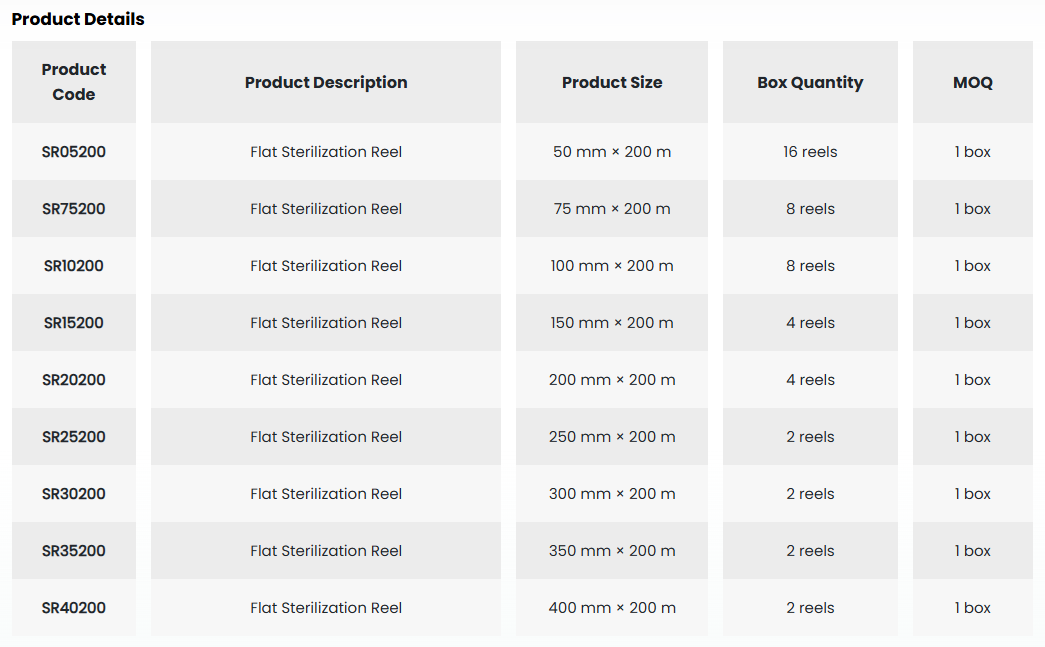

• Designed as a packaging solution for sterilization applications, the Medguard SafeCheck Sterilization Pouch is a microbial barrier system that ensures the sterility of the packed medical device is safely maintained from the moment of sterilization until the time of use.
• It offers a suitable packaging solution for your medical device with a wide range of available sizes.
• It is tear-resistant, aseptic, and easily peelable without leaving any particles.
• It features superior barrier properties.
• The pouch is made of PET/PP copolymer film and 60 GSM or 70 GSM medical kraft paper.
• Steam, Ethylene Oxide (EO), and Formaldehyde (FO) indicator printing options are available.
• The process indicator does not contain lead, heavy metals, or other chemicals harmful to health.
• The pouch meets the performance requirements specified in ISO 11607, ISO 11140-1, and EN 868-5 standards.
• The LOT number, Production Date, and Expiry Date are indicated on the product label.
• The shelf life of the pouch is 5 years.
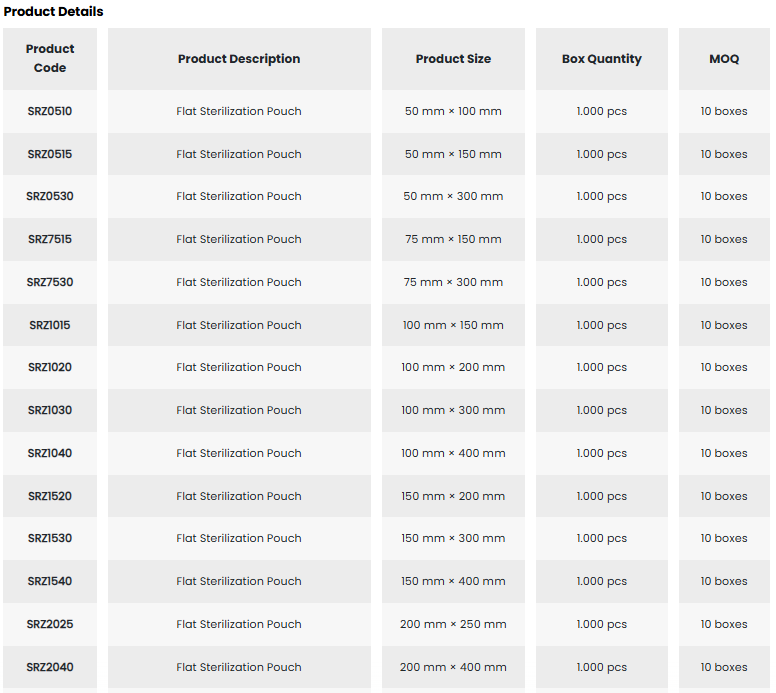
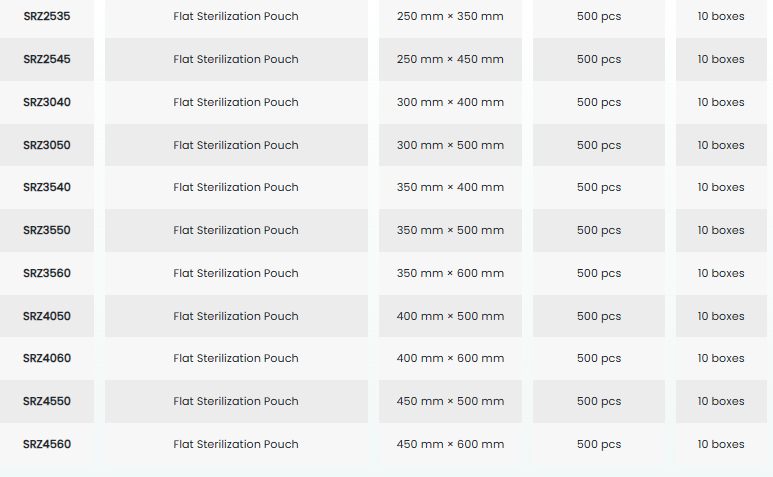

• Designed as a packaging solution for sterilization applications, the Medguard SafeCheck Tyvek® Sterilization Pouch is a microbial barrier system that ensures the sterility of the packed medical device is safely maintained from the time of sterilization until use.
• It is used for low-temperature hydrogen peroxide (H₂O₂) sterilization.
• It is preferred in low-temperature sterilization due to its porous structure.
• The pouch is made of PET/PE laminated film and uncoated DuPont™ Tyvek® web.
• It has superior microbial barrier properties.
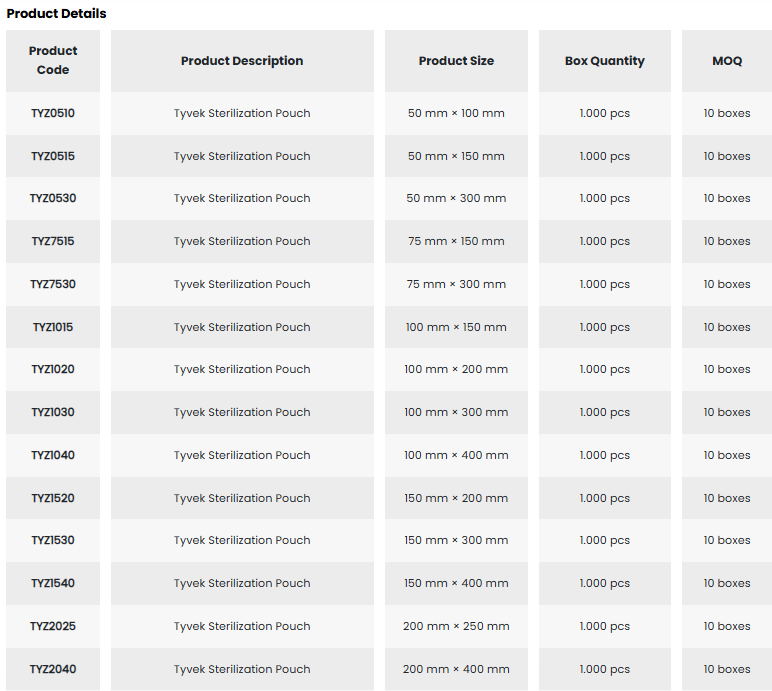
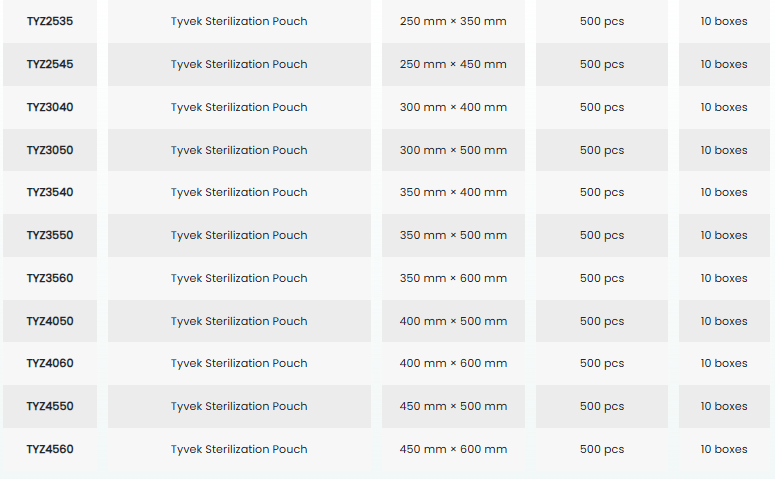
• With its wide range of sizes, it facilitates the selection of appropriate packaging for medical supplies.
• The hydrogen peroxide process indicator is printed with a water-based and non-toxic formulation.
• It features excellent edge welding resistance and a triple-seal structure.
• The product label includes the LOT number, Production Date, and Expiry Date.
• The shelf life of the pouch is 5 years.
Good Manufacturing Practice (GMP) is a system for ensuring that products are consistently produced and controlled according to quality standards. It is designed to minimize the risks involved in any pharmaceutical production that cannot be eliminated through testing the final product.

Request a Quote
